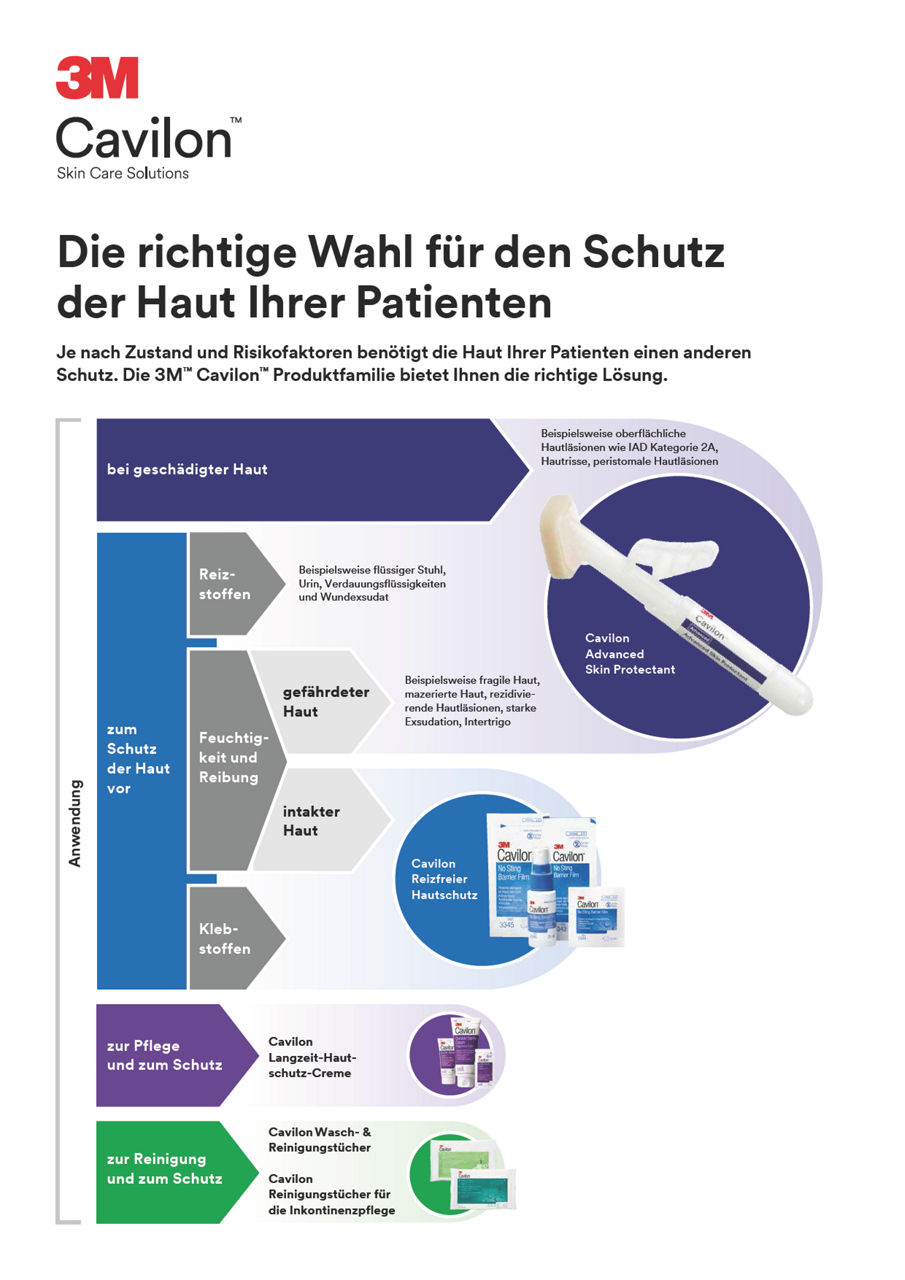
3M™ Cavilon™ Reizfreier Hautschutz
Über das Produkt
3M™ Cavilon™ Reizfreier Hautschutz ist der originale, alkoholfreie, flüssige Barrierefilm, der schnell trocknet und so einen atmungsaktiven, transparenten Schutzfilm auf der Haut bildet.
Produktdetails
- Trocknet schnell und klebt nicht, zur einfachen Anwendung und zur Verbesserung des Patientenwohls⁵
- Alkoholfrei, brennt auch auf gereizter, geschädigter Haut nicht
- Sicher; keine Beeinträchtigung des Heilungsprozesses gereizter, geschädigter Haut³ ⁴
- Barriere gegen Körperflüssigkeiten für verbesserte Kontinenzpflege, die nicht entfernt werden muss
- Schützt nachweislich die Haut bei der Versorgung von Gefäßzugängen⁶. Kompatibel mit Chlorhexidingluconat und Povidon-Iod⁷
- Ermöglicht die Fixierung von Rollenpflastern und Verbänden
- Peel-Open-Verpackung ermöglicht eine sterile Entnahme des Applikators (gilt für 3343)
- Eine bewährte Lösung: über 80 Publikationen belegen die Wirksamkeit und Kosteneffektivität⁸ ⁹
Empfohlene Anwendungen
- Prävention von IAD (Inkontinenz-assoziierter Dermatitis)⁸
- Schutz der Haut um Stomata und Drainagen⁸
- Schutz der Wundumgebung⁸
- Prävention von Hautverletzungen durch medizinische Klebstoffe (MARSI) im Bereich von Gefäßzugängen, unter klebenden Verbänden, Rollenpflastern und Unterdruck-Wundtherapie (NPWT)⁸
- Schutz vor Feuchtigkeit und Reibung ¹⁰ (intertriginöse Dermatitis)
Rechtliches
¹ 2010 Grove et al. 3M White Paper. A Comparison of the Durability of Four Barrier Film Products Over a 72 Hour Period on Human Volunteers. ² CLIN-RPT-FINAL-ICH3-US-05-155867 EM-05-012107 Study to Determine the Ability of the Carbon Black Retention Method to Assess the Durability of Film-Forming Agents, Sponsor Final report (2010). ³ Cameron J, Hoffman D, Wilson J, Cherry G. “Comparison of two peri-wound skin protectants in venous leg ulcers: a randomized controlled trial”. J Wound Care, vol. 14, no. 5, 2005, pp. 233-236. ⁴ Rueda Lopez J, et.al. “A comparative study of a barrier product versus zinc oxide for the treatment of incontinent lesions”. Oral presentation at the World Union Wound Healing Society (WUWHS) Meeting in Paris; 2004. ⁵ CLIN-RPT-FINAL-INV-US-05-289804 EM-05-013869 Study to Assess the Durability of Film-Forming Barriers Using the Activated Carbon Retention Method (2016). ⁶ CLIN-SUPPORT-05-863013 NSBF Clinical Evidence Summary Brochure (2022). ⁷ 3M Data on file. ⁸ CLIN-SUPPORT-05-863013 NSBF Clinical Evidence Summary Brochure (2022). ⁹ CLIN-MISC-US-05-333303 Additional Clinical Evidence for NSBF (2017). ¹⁰ CLIN-MISC-US-05-169008 NSBF Friction Study Grove (2011)Produktspezifikationen
| Marke |
Marke
Cavilon™ |
| VolumeMetric |
VolumeMetric
28 ml |
| Inhalt/Volumen |
Inhalt/Volumen
1 ml, 3 ml, 28 ml |
| Category Name |
Category Name
Reizfreier Hautschutzfilm |
| Applikationssystem |
Applikationssystem
Sonde, Spray, Tuch |
| Inhalt/Volumen (Imperial) |
Inhalt/Volumen (Imperial)
0.034 fl oz (us), 0.101 fl oz (us), 0.947 fl oz (us) |
| DeliverySystem |
DeliverySystem
Sonde |
There was an error processing your request. Please try again later.